|
Founder 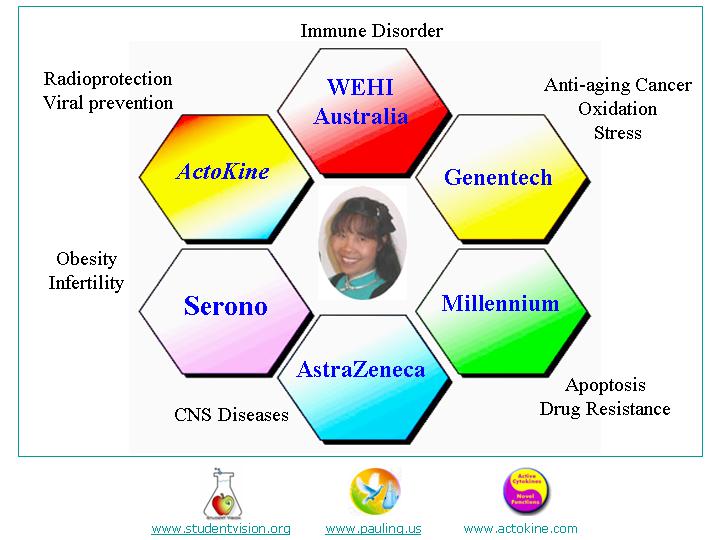
Board of Directors
copyright 2003-2010 |

Founder
Dr Grace Wong CSO, ActoKine Therapeutics
Dr. Grace Wong has worked for Genentech, Millennium, AstraZeneca and Serono on new drug discovery in a variety of therapeutic areas. Dr. Wong earned a PhD at The Walter and Eliza Hall Institute of Medical Research in Australia. Dr. Wong did a postdoc with Dr. David Goeddel at Genentech and advanced basic research discoveries to product development. In 1996, Dr. Wong became the Head of Apoptosis Research at Millennium Pharmaceuticals, worked at AstraZeneca as Section Head of Molecular Genetics and at Serono as Head of Functional Genomics. She has been awarded 13 scholarships from Australian and American organizations and received 5 Recognition Awards from Genentech.
Dr. Wong has been invited to give presentations at 153 international conferences including the Nobel Symposium (Sweden, 1994). She has published 93 papers and filed 28 patents. Some of her publications (3 Nature, 1 Science, 3 Cell, 5 PNAS, and 7 J. Immunol) have received 300-1000 citations. Dr. Wong has founded Actokine Therapeutics for new drug advancements for cancer, obesity, CNS and emerging viral diseases. She also founded Student Vision and the Nobel Pauling Symposia to help scientists of all ages in biotechnology.
Board of Directors
Linus Carl Pauling Jr, MD Board of Director, Student Vision
Dr. Linus Pauling Jr is Chairman and Board of Trustees, Linus Pauling Institute of Science and Medicine. He served in the U.S. Army Air Force during World War II, then did his undergraduate study at Pomona College. He received an M.D. at Harvard University and served as an intern and psychiatric resident at The Queen¡¯s Hospital and The Territorial Hospital in Honolulu. Dr. Pauling has worked as a psychiatrist in private practice and has held positions in mental health organizations, including as president of the Hawaii Mental Health Association. He has also served on the boards of many organizations including the Foundation for Nutritional Advancement, Washington DC and Student Vision, Boston. He is a member of the American Psychiatric Association, Harvard Medical Alumni Association and American Civil Liberties Union.
Hing Leung Sham Ph.D, Sr. VP, Elan Pharmaceuticals (former Abbott)
Before joing Elan as Sr VP, Dr Hing Sham was a Director/Distinguished Research FellowMetabolic Disease Research, Global Pharmaceutical R and D, at Abbott, Dr. Hing Leung Sham, Ph.D received a B.S. in Chemistry from the University of Minnesota, an M.S. in Physical Organic Chemistry from Iowa State University, and a Ph.D in Synthetic Organic Chemistry from the University of Hawaii. He has worked for many years in medicinal chemistry at Abbott. Dr. Sham is the primary inventor of Kaletra (lopinavir/ritonavir), the well-known advanced-generation protease inhibitor for the treatment of HIV infection, and a co-inventor of Norvir (ritonavir), Abbott's first protease inhibitor drug. He currently directs Abbott's medicinal chemistry research efforts in metabolic diseases, including diabetes and obesity. Dr. Sham has published many scientific articles and is a named inventor on 60 issued and 10 pending U.S. patents. He has served as an editor and reviewer, and is a member of many scientific associations. Dr. Sham received the 2003 Heroes of Chemistry Award from the American Chemical Society (for his Kaletra discovery), the National Inventor of the Year award from the Intellectual Property Owners Association (for Norvir), and twice the Inventor of the Year award from the Intellectual Property Law Association of Chicago (for Norvir and Kaletra).
Claude G. Biava MD, Chief Medical Officer, ActoKine Therapeutics.
Dr. Biava has more than 30 years of experience in pre-clinical and clinical research studies. From 1968-1971, as the Director of the Department of Pathology and Toxicology in the Experimental Therapy Division at Abbott Laboratories, he was responsible for pre-clinical safety evaluation of new drug candidates and their approval for clinical testing, preparation of data for submission of INDs and NDAs. As Vice President for R&D and Clinical Research in the Hospital Products Division at Abbott Laboratories from 1971-1973, he was responsible for R&D and clinical studies of hospital products, including anesthetics, intravenous therapeutics, urological products, and intraperitoneal dialysis kits. From 1973 to 1993, he was an Associate Professor and Director of the Laboratory of Ultrastructural and Renal Pathology Services at the University of California, San Francisco Medical Center. Dr. Biava received his M.D. from Louvain University in Belgium and completed his residency as a Royal Canadian Research Fellow at Queen¡¯s University in Kingston, Ontario. He is the author of more than 50 publications.
Robert Allen Lewis MD,Former Sr VP and Site Head for the Aventis Pharmaceuticals, USA
Dr. Robert A. Lewis was educated at Yale (BA, chemistry, 1967), the University of Rochester (MD, 1971), and Harvard (Internship, Residency in Pediatrics, Children's Hospital Med Center; Fellowship in Immunology, Harvard Med. School). After two years as a staff rheumatologist and allergist in the US Air Force, Dr. Lewis joined the faculty of Harvard Med. School, where he remained for over a decade, conducting research on mast cells, prostaglandins and leukotrienes, in collaboration with his department chairman, Dr. K. Frank Austen, a colleague from pulmonary medicine, Dr. Jeffrey Drazen, the Sheldon Emory Professor of Chemistry at Harvard University, Dr. E J Corey, and a number of talented post-doctoral fellows and students. In 1986, Dr. Lewis left Harvard to join Syntex Corporation as director of basic research, from which he advanced to become President of Discovery Research; there, he and his colleagues developed several drugs, including myocophenolate mofetil for prevention of acute transplant rejection, and gancyclovir and valgancyclovir for therapy of cytomegalovirus infections. In 1995, approximately a year after Roche Holdings acquired Syntex, Dr. Lewis moved to Cell Therapeutics in Seattle, as Chief Scientific Officer, where, with his colleagues, he developed polyglutamic acid polymer conjugates with taxanes and other hydrophobic cancer drugs and cloned many of the critical human enzymes involved in the turnover of phospholipids, with focus on their effects in oncogenesis. There, with colleagues, he also began exploring gene expression during T-cell subtype differentiation. In 2000, Dr. Lewis moved to Aventis Pharmaceuticals at the Bridgewater, NJ campus, to create and direct a center for expertise in immunology research, termed the Immunology Platform, which has become the center for expression profiling of human immunocytes at Aventis. For the past six months, Dr. Lewis has also been acting Sr VP and Site Head for the Aventis US Research Site. Dr. Lewis is the author or coauthor of 160 scientific papers and book chapters on cell biology and biochemistry in immediate hypersensitivity and related disorders. He has served on the faculties of Harvard, Stanford, and UCSF medical schools and has been an invited speaker at numerous national and international meetings over the past 25 years, including the invited professorial lectureship of the Japanese Society of Allergology in 1983.
Martin A. Wasserman, Ph.D., (former Aventis, GSK, BMS, Roche)
Dr. Wasserman is an internationally recognized and accomplished biomedical scientist and pharmaceutical executive with 35+ years of industrial research and development experience. He retired voluntarily as Senior Vice President of Discovery Research and Chief Scientific Officer at AtheroGenics, Inc., in Alpharetta, GA, where he remains a Consultant. Prior to joining AtheroGenics, Dr. Wasserman was Vice President and Senior Distinguished Scientist in the Respiratory and Rheumatoid Arthritis Disease Group within the U.S. Drug Innovation and Approval Organization (R&D) at Sanofi-Aventis Pharmaceuticals (1995-2001) and, earlier, he was a valued and productive colleague at Pfizer (1972-1981), GlaxoSmithkline (1981-1988), Bristol-Myers Squibb (1989-1991) and Hoffmann-La Roche (1992-1995). He has a B.S. degree in Pharmacy from The Ernest Mario School of Pharmacy at Rutgers University (formerly Rutgers University College of Pharmacy) in NJ and was awarded M.A. and Ph.D. degrees in Pharmacology and Toxicology from the University of Texas Medical Branch in Galveston, TX (from which he was also honored with the Distinguished Alumnus Award in 1986). His areas of biomedical R&D expertise and experience encompass a broad range of therapeutic disciplines including: bronchopulmonary, allergy, cardiovascular, inflammation, immunology, renal, oncology, endocrinology, and gastroenterology. His academic distinctions are numerous. Dr. Wasserman was appointed Adjunct Professor of Medicine at Emory University in 2002 and formerly held four additional Adjunct Professorships: The University of Medicine and Dentistry of New Jersey, The Ernest Mario School of Pharmacy at Rutgers University, Philadelphia College of Pharmacy and Sciences, and Seton Hall University School of Graduate Medical Education. He is on the Editorial Boards of several journals, e.g., The Journal of Pharmacology and Experimental Therapeutics and the Yearbook of Pharmacology. He has published 84 peer-reviewed research papers in international scientific journals as well as authoring or co-authoring 137 abstracts and 23 book chapters and scientific reviews. Additionally, he has been a Visiting Professor at: Boston University School of Medicine (1985), Vanderbilt University School of Medicine (1986, 1987), Medical College of Georgia (2004) and Georgia Institute of Technology (2004).
Scientific Advisory Committee
Jeffrey Browning PhD, Distinguished Scientist, Biogen
Linda Burkly PhD, Distinguished Investigator, Biogen
Diane Pennica PhD, Senior Scientist, Genentech
Napoleone Ferrara PhD, Staff Scientist, Genentech
David Leung PhD, Director of Molecular Biology, Cell Therapeutics
Pat Gray PhD VP of Research, MacroGenics
Kuldeep Neote PhD, Director of Research, Eli Lilly
Reinhard Ebner PhD, Scientific Director, Avalon Pharmaceuticals.
Jim Strickler PhD, Director of Suntory Pharmaceutical Research Labs
Steve Arkinstall PhD, VP of Research, Head of SRBI, Serono (Boston)
Tim Wells PhD, Head of Drug Discovery, Former Serono (Geneva)
Shi-Chung Ng PhD, former Abbott
Alexander Sasha Kamb PhD, Oncology, Amgen
David Merberg, PhD, Director of Informatics at Cell Signaling Technology
Rick Bott PhD, Staff Scientist, Genencor (Danisco)
Dr. Zhijian Lu PhD Associate Director, Wyeth
Gordon Vehar PhD VP of Research, Raven Biotechnologies, SSF, CA, USA
David He, PhD Abbott
Scott Wadsworth, PhD Johnson & Johnson
Home | Mission | Registration | Sponsors | Job Strategies | News, Events, Contact
Biotech Booth | Event Photos | Nobel Day | Dr. Linus Pauling | Who's Who
Pauling Biotech Symposium | Symposium Agenda | Symposium Speakers | Symposium Testimonials
Founder & Board of Directors | Scientific Advisory Committee | Scholarship Application
copyright 2003-2010